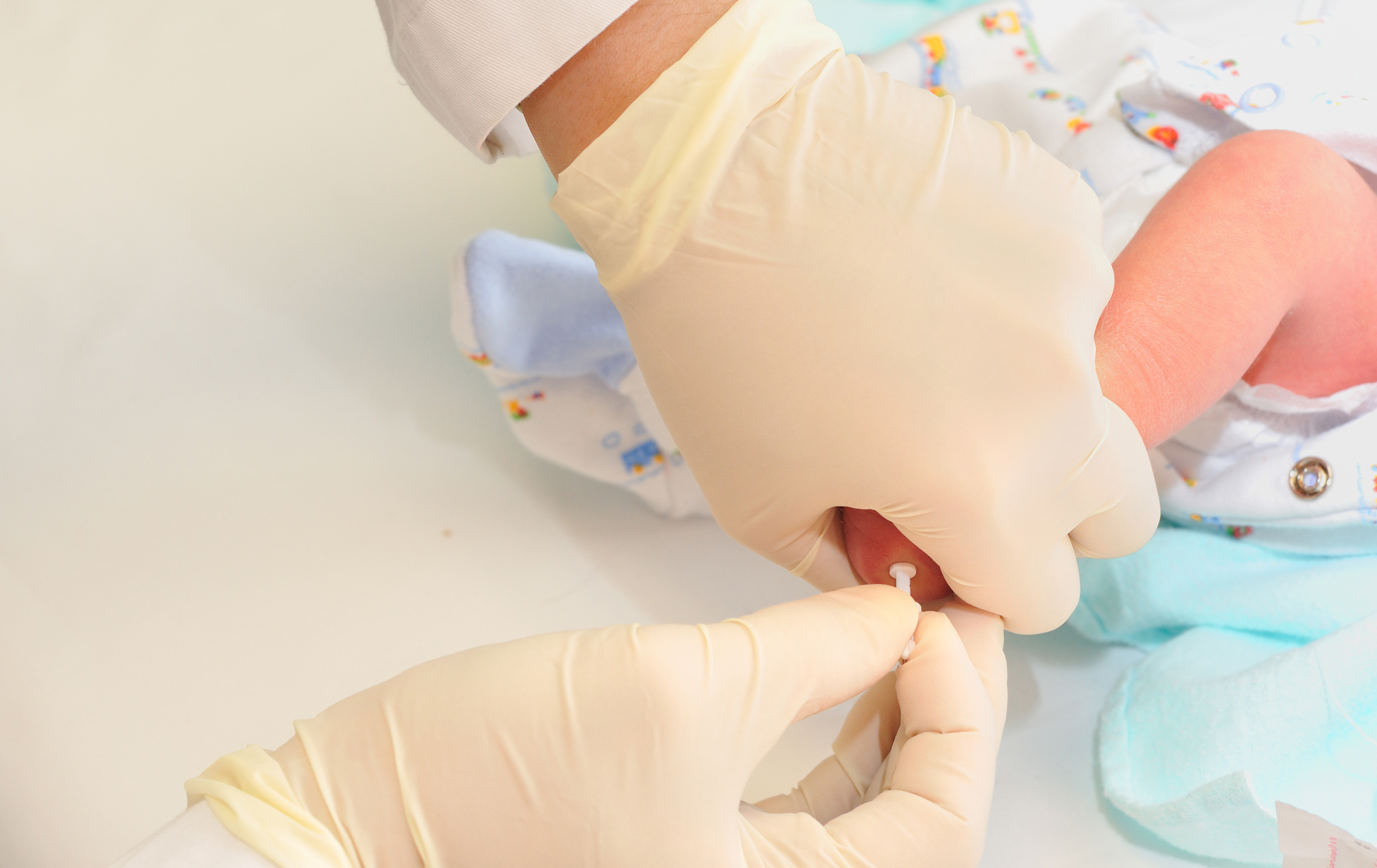
When you have a day-old baby, a nurse or a phlebotomist performs a heel stick to take a few drops of blood from your infant and sends it off to a state lab for a battery of tests. Most of the time, you never hear about the results because your child is fortunate enough to not have a rare disease, such as cystic fibrosis or sickle cell disease, or any of the dozens of conditions for which most states screen. You, as a parent, may not even remember hearing about newborn screening.
Newborn screening is mandatory in most states, including California, unless parents refuse for religious or other reasons. Screening is generally accepted because it is only performed for conditions where measures are available to save the baby’s life or mitigate the harms of such conditions, if found early enough—and where, without screening, the disease would not be clinically evident and so would likely not be recognized until too late. However, now that scientists have developed methods for sequencing the entire genome, what would happen if states began incorporating genome sequencing to find out more about baby’s health? How would that work? What should parents learn about their baby’s genome? What shouldn’t they?
Steven Brenner, a professor in the Department of Plant and Microbial Biology, is part of a national consortium of researchers and doctors studying the ins and outs of potentially using genome sequencing for newborn health screenings and beyond.
Called NSIGHT, the consortium includes four NIH grants and spans multiple institutions, including 4 lead institutions:
- University of California, San Francisco (UCSF)
- University of North Carolina School of Medicine
- Brigham and Women’s Hospital/ Boston Children’s Hospital and Baylor College of Medicine
- University of California San Diego Rady Children’s Institute for Genomic Medicine
The group published an overview of their current and future research in the journal Pediatrics last month. Their studies are working to address using genome sequencing in three clinical scenarios:
- Screening: to test healthy newborns for preventable or treatable conditions that genetic sequencing could detect or help confirm.
- Diagnostic: to find the specific genetic causes of congenital anomalies or unexplained illnesses in babies admitted to neonatal intensive care units.
- Predictive: to explore the entire genome of the child, as a resource for health care throughout the course of the child’s life.
In collaboration with researchers at UCSF, the California Department of Public Health, and Tata Consultancy Services, Brenner is a part of the group working on the screening scenario, with research in computational genomics. The group is creating the tools needed to analyze the genome for the dozens of metabolic disorders that are now a part of California’s newborn screening program. The state has archived residual dried blood spot samples from infants since 1982. The group focused on a subset of the archive—samples collected from approximately 4 million babies from 2005-2013—analyzing blood from the 1,500 babies in that group who had an inherited metabolic disorder identified by screening. At first it was not clear that enough intact DNA could be obtained from the tiny dried blood spots punched out from the archived filter paper, but as Jennifer Puck of UCSF said, “It is remarkable that the sequence quality obtained from this material was equivalent to sequences from fresh blood samples.” Puck is co-principal investigator for the grant and leader of the UCSF DNA extraction laboratory.
From all this material and multiple DNA databases that serve as a reference tool, Brenner’s group aims to sort out the gene variants involved in the disorders and assess what roles they play. “The metabolic disorders are amongst the best understood genetic diseases, which makes them a promising set of disorders to explore with genomic sequencing,” said Brenner. “Our results are showing both unexpected limitations as well as potentially lifesaving applications of the genomic technology in screening.”
This NSIGHT consortium will work with parents and conduct genomic sequencing on newborns to develop evidence that may support guidelines for whether and how this new technology could be effectively and appropriately incorporated into newborn screening or the care of newborns.
In addition to the lead sites, NSIGHT includes researchers and administrators from the National Institutes of Health (the National Human Genome Research Institute, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Center for Advancing Translational Sciences); University of California, Berkeley; RTI International; American College of Medical Genetics and Genomics; Harvard Medical School; University of Illinois-Chicago School of Public Health; California Department of Public Health; Oregon Health & Sciences University; Tata Consultancy Services; and University of Washington.
Related Information
